We do not offer international shipping to countries within the European Union
We can only ship non-hazardous products to Rest of World





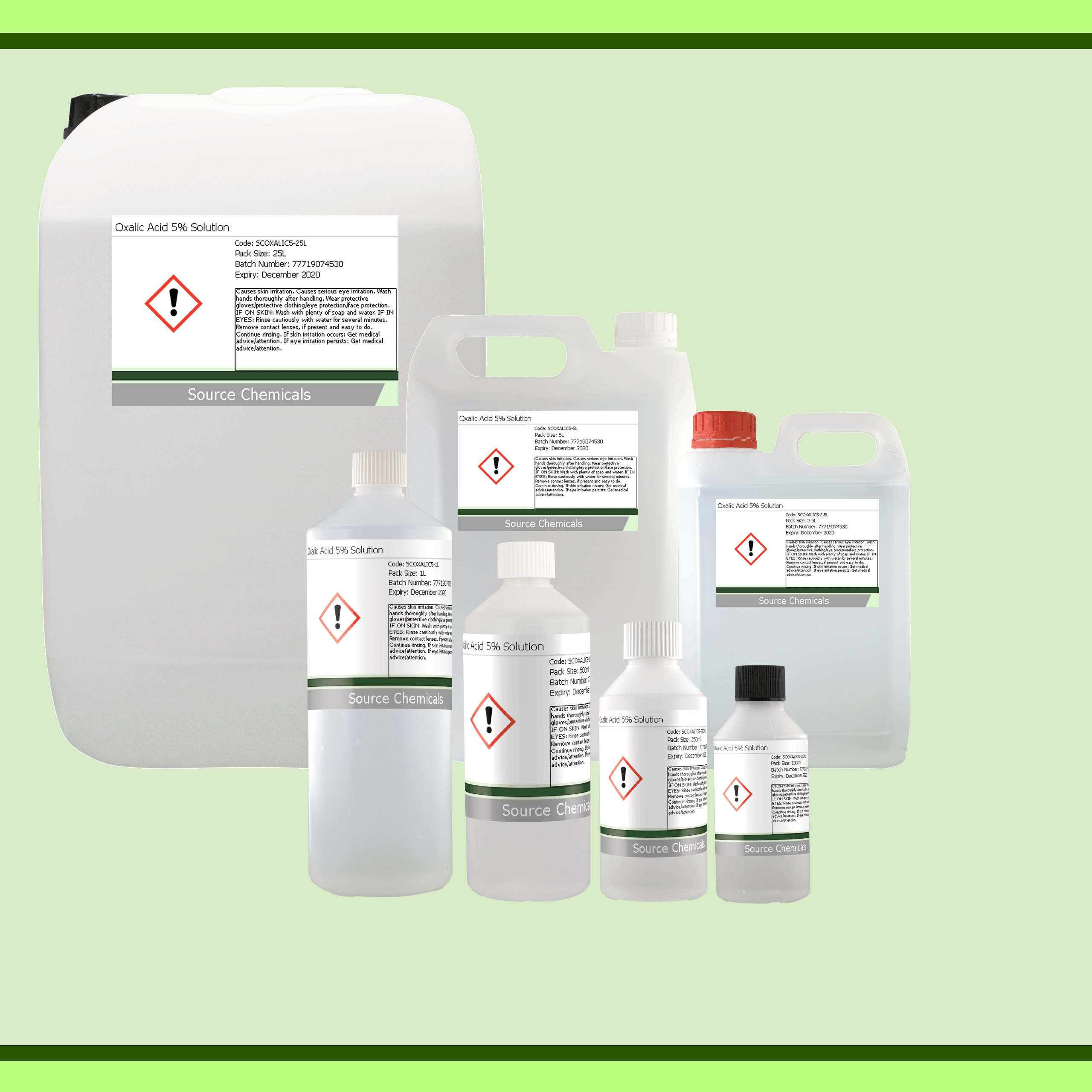
Compare all Pack Sizes
Unfortunately, we are not currently accepting orders for Northern Ireland, Jersey and Guernsey.
We can only ship to the following postcodes where a Royal Mail postal option has been chosen at checkout.
AB, DD, DG3, DG4, DG6 - DG9, EH, FK, G, HS, IM, IV, KA, KW, KY, ML, PA, PH, PO30 - PO41, TD, TR21 - TR25, ZE
A 5% Solution of Oxalic Acid.
Oxalic acid is an essential household chemical that can be used, like many acids, as a cleaner for various things; for example, as a rust remover, a cleaning agent, on wood work as a stain lifter, as a bleaching agent, and many more.
Contains:
Oxalic Acid: 5%
Distilled Water: 95%
Hazard Phrases: Causes severe skin burns and eye damage. Causes serious eye damage.
Prec Phrases: Do not breathe dust/fume/gas/mist/vapours/spray. Wear protective gloves/protective clothing/eye protection/face protection. Dispose of contents/container to



























We can also accept a bank transfer (UK Only)